University of Colo. making hydrogen with solar
 Fuel cells have long promised to be the electricity producer of the future, but there have been some nagging problems.
Fuel cells have long promised to be the electricity producer of the future, but there have been some nagging problems.
One of the biggest is there’s not enough hydrogen to make them viable. Sure there’s enough hydrogen fixed in water, but splitting water into hydrogen and oxygen takes a lot of energy. But research at the University of Colorado, Boulder (CU) is using concentrated sunlight to split brackish water into hydrogen and oxygen, and it’s expected to meet the Department of Energy’s (DOE’s) cost targets for marketable hydrogen.
Currently the most cost-effective means of producing hydrogen is steam-methane reforming of natural gas, but it relies on fossil fuels and releases carbon dioxide into the atmosphere. By splitting water into its constituent elements, even brackish water such as seawater, there’s little or no resulting pollution. The hydrogen and oxygen can be stored and transported or used directly in a fuel cell to produce electricity.
A DOE-commissioned report concluded CU’s method of splitting water with sunlight is the only approach among eight competing technologies that is projected to meet DOE’s future cost targets. It’s expected to cost $6 dollars per kilogram by 2015 and $2 to $3 dollars per kg by 2025, according to a CU press release. The research, which uses thin-film nanotechnology is being led by Alan Weimer, H.T. Sears Memorial Professor in Chemical and Biological Engineering at CU.
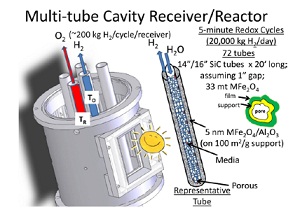
“Brackish water enters and is evaporated to a distilled water stream and a concentrate stream,” Weimer said. “The evaporated water is more than is needed for supplying the solar water splitting reactor, and so it can be used for agriculture or drinking, etc. The heat associated with the released O2 [i.e., oxygen gas] and H2 [i.e., hydrogen gas] is used to drive the multiple-effect evaporator.” Remaining effluents would be minimal, but would have to be disposed of.
The prototype device, tested at the National Renewable Energy Laboratory, concentrates sunlight using mirrors and heats it to temperatures as high as 2,640 degrees Fahrenheit—482 degrees lower than other water splitting technologies. The reaction of a thin-film metal ferrite coating with a reactive substrate contained in a solar receiver splits water into hydrogen and oxygen.
Weimer envisions the device for distributed energy production.
“I think the best approach is a dish based system for distributed H2 for a small fueling station or home fueling to complement the roll out of fuel cell vehicles,” he said. “A large dish could certainly make 1 to several kg of H2 per day, and since a fuel cell car can get 400 miles on 5 kg, such a size could be used for home fueling.”
The process could also be used to produce electricity via a steam turbine in a subsequent step, according Weimer.
“The water splitting runs at higher temperature, so it may be possible to use the waste heat to drive a steam generator like a solar tower process,” he said.



